NCERT Solutions for Class 10 Science Chapter 1 Chemical Reactions and Equations
NCERT Solutions for Class 10 Science Chapter 1 Intext Questions
Page Number: 6
Question 1
Why should a magnesium ribbon be cleaned before burning in air ?
Answer:
Magnesium gets covered with a layer of magnesium oxide when kept in air for a long time. This layer hinders the burning of magnesium. Hence, it is to be cleaned before burning.
Question 2
Write the balanced equation for the following chemical reactions.
(i) Hydrogen + Chlorine → Hydrogen chloride
(ii) Barium chloride + Aluminium sulphate → Barium sulphate + Aluminium chloride
(iii) Sodium + Water → Sodium hydroxide + Hydrogen
Answer:
(i) H2 + Cl2 → 2HCl
(ii) 3 BaCl2 + Al2(SO4)3 → BaSO4 + 2 AlCl3
(iii) 2Na + 2H2O → 2NaOH + H2↑
Question 3
Write a balanced chemical equation with state symbols for the following reactions :
(i) Solutions of barium chloride and sodium sulphate in water react to give insoluble barium sulphate and the solution of sodium chloride.
(ii) Sodium hydroxide solution (in water) reacts with hydrochloric acid solution (in water) to produce sodium chloride solution and water.
Answer:
(i) BaCl2 (aq) + Na2SO4 (aq) → BaSO4(s) + 2NaCl (aq)
(ii) NaOH (aq) + HCl(aq) → NaCl(aq) + H2O(l)
Page Number: 10
Question 1
A solution of a substance ‘X’ is used for white washing.
(i) Name the substance ‘X’ and write its formula.
(ii) Write the reaction of the substance ‘X’ named in (i) above with water.
Answer:
(i) The substance whose solution in water is used for white washing is calcium oxide (or quick lime). Its formula is CaO.
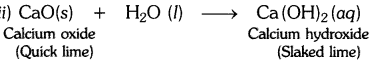
Question 2
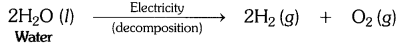
Why is the amount of gas collected in one of the test tubes in text book Activity 1.7 (i.e., electrolysis of water) double of the amount collected in the other? Name this gas. [CBSE 2015 (Delhi)]
Answer:
In Activity 1.7, water is electrolysed to give H2 gas at one electrode and O2 gas at the other electrode.
2H2O(l) → 2H2(g) + O2(g)
Thus two molecules of water on electrolysis give two molecules of hydrogen gas and one molecule of oxygen gas or in other words the amount of hydrogen gas collected would be double than that of oxygen gas.
Page Number: 13
Question 1
Why does the colour of copper sulphate solution change when an iron nail is dipped in it ?
OR
An iron nail is dipped in the solution of copper sulphate for about 30 minutes. State the change in colour observed. Give reason for the change. [CBSE 2015 (Delhi)]
Answer:
When an iron nail is dipped in copper sulphate solution, the displacement reaction takes place. The colour of copper sulphate solution fades due to the formation of light green solution of iron sulphate.
![]()
Question 2
Give an example of a double displacement reaction other than the one given in Activity 1.10 (NCERT Text Book).
Answer:
Sodium hydroxide and hydrochloric acid react to form sodium chloride and water.
![]()
Question 3
Identify the substances that are oxidised and the substances which are reduced in the following reactions.
(i) 4Na(s) + O2(g) → 2Na2O(s)
(ii) CuO (s) + H2(g) → Cu (s) + H2O(l)
Answer:
(i) Substances oxidised is Na as it gains oxygen and oxygen is reduced.
(ii) Substances reduced is Cu as hydrogen is oxidised as it gains oxygen.
NCERT Solutions for Class 10 Science Chapter 1 Textbook Chapter End Questions
Question 1
Which of the statements about the reaction below are incorrect ?
2 PbO(s) + C(s) → 2Pb (s) + CO2(g)
(a) Lead is getting reduced.
(b) Carbon dioxide is getting oxidised.
(c) Carbon is getting oxidised.
(d) Lead oxide is getting reduced.
(i) (a) and (b)
(ii) (a) and (c)
(iii) (a), (b) and (c)
(iv) All
Answer:
(i) (a) and (b)
Question 2
Fe2O3 + 2Al → Al2O3 + 2Fe
The above reaction is an example of a
(a) combination reaction
(b) double displacement reaction
(c) decomposition reaction
(d) displacement reaction
Answer:
(d) Displacement reaction.
Question 3
What happens when dilute hydrochloric acid is added to iron filings ? Tick the correct answer :
(a) Hydrogen gas and iron chloride are produced.
(b) Chlorine gas and iron hydroxide are produced.
(c) No reaction takes place.
(d) Iron salt and water are produced.
Answer:
(a) Hydrogen gas and iron chloride are produced.
Question 4
What is a balanced chemical equation ? Why should chemical equations be balanced ?
Answer:
A balanced chemical equation has an equal number of atoms of different elements in the reactants and products.
The chemical equations should be balanced to satisfy the law of conservation of mass.
Question 5
Translate the following statements into chemical equations and then balance them.
(a) Hydrogen gas combines with nitrogen to form ammonia.
(b) Hydrogen sulphide gas burns in air to give water and sulphur dioxide.
(c) Barium chloride reacts with aluminium sulphate to give aluminium chloride and a precipitate of barium sulphate.
(d) Potassium metal reacts with water to give potassium hydroxide and hydrogen gas.
Answer:
(a) 3H2 (g) + N2 (g) → 2NH3 (g)
(b) H2S (g) + 3O2 (g) → SO2 (g) + 2H2O(l)
(c) 3BaCl2 (aq) + Al2(SO4)3 (aq) → 2AlCl3 (aq) + 3BaSO4 ↓(s)
(d) 2K (s) + 2H2O (l) → 2KOH (aq) + H2 (g)
Question 6
Balance the following chemical equations :
(a) HNO3 + Ca (OH)2 → Ca (NO3)2 + H2O
(b) NaOH + H2SO4 → Na2SO4 + H2O
(c) NaCl + AgNO3 → AgCl + NaNO3
(d) BaCl2 + H2SO4 → BaSO4 + HCl
Answer:
(a) 2HNO3 + Ca(OH)2 → Ca(NO3)2 + 2H2O
(b) 2NaOH + H2SO4 → Na2SO4 + 2H2O
(c) NaCl + AgNO3 → AgCl + NaNO3
(d) BaCl2 + H2SO4 → BaSO4 + 2HCl
Question 7
Write the balanced chemical equations for the following reactions :
(a) Calcium hydroxide + Carbon dioxide → Calcium carbonate + Water
(b) Zinc + Silver nitrate → Zinc nitrate + Silver
(c) Aluminium + Copper chloride → Aluminium chloride + Copper
(d) Barium chloride + Potassium sulphate → Barium sulphate + Potassium chloride
Answer:
(a) Ca (OH)2 + CO2 → CaCO3 + H2O
(b) Zn + 2AgNO3 → Zn(NO3)2 + 2 Ag
(c) 2Al + 3 CuCl2 → 2AlCl3 + 3 Cu
(d) BaCl2 + K2SO4 → BaSO4 + 2KCl
Question 8
Write the balanced chemical equation for the following and identify the type of reaction in each case :
(a) Potassium bromide (aq) + Barium iodide (aq) → Potassium iodide (aq) + Barium
(b) Zinc carbonate(s) → Zinc oxide (s) + Carbon dioxide (g) bromide(s)
(c) Hydrogen (g) + Chloride (g) → Hydrogen chloride (g)
(d) Magnesium (s) + Hydrochloric acid (aq) → Magnesium chloride (aq) + Hydrogen (g)
Answer:
(a) 2KBr (aq) + Bal2(aq) → 2Kl(aq) + BaBr2(s)
Type : Double displacement reaction
(b) ZnCO3 (s) → ZnO (s) + CO2 (g)
Type : Decomposition reaction
(c) H2 (g) + Cl2 (g) → 2HCl(g)
Type : Combination reaction
(d) Mg (s) + 2HCl (aq) → MgCl2 (aq) + H2 (g)
Type : Displacement reaction
Question 9
What does one mean by exothermic and endothermic reactions ? Give examples.
Answer:
Exothermic reactions : Those reactions in which heat is evolved are known as exothermic reactions. An exothermic reaction is indicated by writing “+ Heat”on the products side of an equation.
Example :
(i) C (s) + O2 (g) → CO2 (g) + Heat
(ii) N2 (g) + 3H2 (g) → 2NH3 (g) + Heat
Endothermic reactions : Those reactions in which heat is absorbed are known as endothermic reactions. An endothermic reaction is usually indicated by writing “Heat” on the product side of a chemical equation.
Examples :
(i) C (s) + 2S (s) → CS2 (l) – Heat
(ii) N2 (g) + O2 (g) → 2NO(g) – Heat
Question 10
Why is respiration considered an exothermic reaction ? Explain.
Answer:
Respiration is an exothermic process because during respiration glucose combines with oxygen in the cells of our body to form carbon dioxide and water along with the production of energy.
![]()
Question 11
Why are decomposition reactions called the opposite of combination reactions? Write equations for these reactions.
Answer:
In a decomposition reaction, a single compound breaks down to produce two or more simpler substances.
For example:
While, in a combination reaction, two or more substances simply combine to form a new substance.
For example:
![]()
Question 12
Write one equation each for the decomposition reactions where energy is supplied in the form of heat, light or electricity.
OR
Decomposition reactions require energy either in the form of heat or light or electricity for breaking down the reactants. Write one equation each for decomposition reactions where energy is supplied in the form of heat, light and electricity. [CBSE 2015 (Delhi)]
Answer:
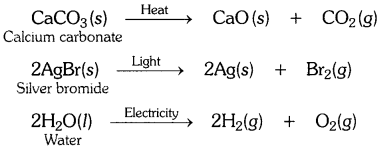
Question 13
What is the difference between displacement and double displacement reactions? Write equations for these reactions.
Answer:
In displacement reactions, a more reactive metal displaces a less reactive metal from its solution. For example,
Fe(s) + CuSO4(aq) → Cu(s) + FeSO4(aq)
This is a displacement reaction where iron displaces copper from its solution.
In double displacement reactions, two reactants in solution exchange their ions. For example,
AgNO3(aq) + NaCl (aq) → AgCl(s) + NaNO3 (aq)
This is a double displacement reaction where silver nitrate and sodium chloride exchange Cl– and NO3– ions between them.
Question 14
In the refining of silver, the recovery of silver from silver nitrate solution involved displacement by copper metal. Write down the reaction involved.
Answer:
![]()
Question 15
What do you mean by a precipitation reaction ? Explain by giving examples.
Answer:
A reaction in which an insoluble solid called precipitate is formed that separates from the solution is called a precipitation reaction.
Example : When a solution of iron (III) chloride and ammonium hydroxide are mixed, a brown precipitate of iron (III) hydroxide is formed.
![]()
Question 16
Explain the following in terms of gain or loss of oxygen with two examples each:
(a) Oxidation and
(b) Reduction.
Answer:
(a) Oxidation : The addition of oxygen to a substance is called oxidation.
Example :
(i) S(s) + O2(g) → SO2(g) (Addition of oxygen to sulphur)
(ii) 2Mg(s) + O2 (g) → 2MgO(s) (Addition of oxygen to magnesium)
(b) Reduction : The removal of oxygen from a substance is called reduction.
Example: (i) CuO + H2 \(\underrightarrow { Heat }\) Cu + H2O
Here, copper oxide is being reduced to copper because oxygen gets removed from copper oxide.
(ii) ZnO + C → Zn + CO
Here, zinc oxide is being reduced to zinc because oxygen gets removed from zinc oxide.
Question 17
A shiny brown coloured element ‘X’ on heating in air becomes black in colour. Name the element ‘X’ and the black coloured compound formed.
Answer:
Element ‘X’ is copper (Cu).
The black coloured compound is copper oxide (CuO). The reaction involved is
![]()
Question 18
Why do we apply paint on iron articles ?
Answer:
Paint does not allow iron articles to come in contact with air, water and saves iron articles from damage due to rusting.
Question 19
Oil and fat containing food items are flushed with nitrogen. Why ?
Answer:
To keep food items fresh and save from getting oxidised, food items are flushed with nitrogen.
Question 20
Explain the following terms with one example each (a) Corrosion, (b) Rancidity.
Answer:
(a) Corrosion : It is the process in which metals are eaten up gradually by the action of air, moisture or a chemical (such as an acid) on their surface.
Example : When iron is exposed to moist air for a long period of time, its surface acquires a coating of a brown, flaky substance called rust. Rust is mainly hydrated iron (III) oxide [Fe2O3.xH20].
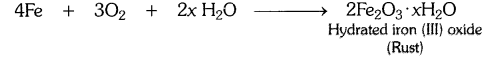
(b) Rancidity : The condition produced by aerial oxidation of fats and oils in foods marked by unpleasant smell and taste is called rancidity.
Rancidity spoils the food materials prepared in fats and oils which have been kept for a considerable time and makes them unfit for eating.
Rancidity can be prevented by adding anti-oxidants to foods containing fats and oils. It can also be prevented by flushing fat and oil containing foods with nitrogen before sealing.
NCERT Solutions for Class 10 Science Chapter 1 Chemical Reactions and Equations
NCERT Solutions for Class 10 Science (Chemistry) Chapter 1 Chemical Reactions and Equations are part of NCERT Solutions for Class 10 Science. Here we have given Class 10 Science NCERT Solutions Chapter 1.
Question 1
Which of the given statements about the reaction below are incorrect?
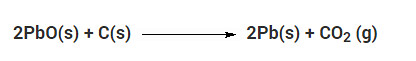
a. Lead is getting reduced
b. Carbon dioxide is getting oxidised
c. Carbon is getting oxidised
d. Lead oxide is getting reduced
(i) (a) and (b)
(ii) (a) and (c)
(iii) (a), (b) and (c)
(iv) all
Solution:
(i) (a) and (b)
Question 2
![]()
The above reaction is an example of a
(i). Combination reaction
(ii). Double displacement reaction
(iii). Decomposition reaction
(iv). Displacement reaction
Solution:
(iv) Displacement reaction
Question 3
What happens when dilute hydrochloric acid is added to iron fillings? Tick the correct answer.
a. Hydrogen gas and iron chloride are produced
b. Chlorine gas and iron hydroxide are produced
c. No reaction takes place
d Iron salt and water are produced
Solution:
Hydrogen gas and iron chloride are produced
Question 4
What is a balanced chemical equation? Why should chemical equations be balanced?
Solution:
An equation for a chemical reaction is an equation in which the number of atoms for each element in the reaction and the total charge is the same for both the reactants and the products. In other words, the mass and the charge are balanced on both sides of the reaction.
Question 5
Translate the following statements into chemical equations and then balance them.
a. Hydrogen gas combines with nitrogen to form ammonia
b. Hydrogen sulfide gas burns in air to give water and sulfur dioxide
c. Barium chloride reacts with Aluminium sulfate to give Aluminium chloride and a precipitate of barium sulphate
d. Potassium metal reacts with water to give a potassium hydroxide and hydrogen gas
Solution:
a. 3H2 + N2 → 2NH3
b. 2H2S + 3O2 → 2H2O + 2SO2
c. 3BaCl2 + Al2(SO4)3 → 2AlCl3 + 3BaSO4
d. 2K + 2H2O → 2KOH + H2
Question 6
Balance the following chemical equations.
a. HNO3 + Ca(OH)2 → Ca(NO3)2 + H2O
b.NaOH + H2SO4 → Na2SO4 + H2O
C. NaCl + AgNO3 → AgCl + NaNO3
d. BaCl2 + H2SO4 → BaSO4 + HCl
Solution:
a. 2HNO3 + 2Ca(OH)2 → Ca(NO3)2 + 2H2O
b. 6NaOH + 3H2SO4 → 3Na2SO4 + 6H2O
C. NaCl + AgNO3 → AgCI + NaNO3
d. BaCl2 + H2SO4 → BaSO4 + 2HCl
Question 7
Write the balanced chemical equations for the following reactions:
(a) Calcium hydroxide + Carbon dioxide → Calcium carbonate + Water(b) Zinc + Silver nitrate → Zinc nitrate + Silver(c) Aluminium + Copper chloride → Aluminium chloride + Copper(d) Barium chloride + Potassium sulphate → Barium sulphate + Potassium chlorideAnswer:(a) Ca (OH)2 + CO2 → CaCO3 + H2O(b) Zn + 2AgNO3 → Zn(NO3)2 + 2 Ag(c) 2Al + 3 CuCl2 → 2AlCl3 + 3 Cu(d) BaCl2 + K2SO4 → BaSO4 + 2KCl
Question 8
Write the balanced chemical equation for the following and identify the type of reaction of each case.
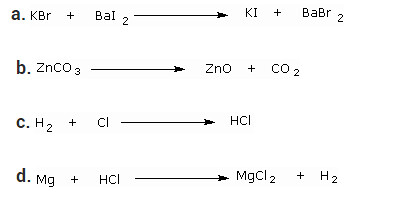
Solution:
a. 2KBr + BaI2 → 2KI + BaBr2 — Double Displacement reaction
b. ZnCO3 → ZnO + CO2 — Decomposition reaction
c. H2 + Cl2 → 2HCl — Combination reaction
d. Mg + 2HCl → MgCl2 + H2 — Displacement reaction
Question 9
What is meant by exothermic and endothermic reactions? Give examples.
Solution:
An exothermic reaction is one that releases heat.
If 1 mole of N2 molecule reacts with 1 mole of O2 molecule, the heat of 184 KJ has to be supplied to initiate the reaction to give 1 mole of NO molecule. This means that the bonds between N – N and O – O are so strong that they do not break easily. N2 has triple covalent bond between the two N atoms. O2 has a double covalent bond. Thus energy has to be put into the reaction to break the strong bonds. Thus the above reaction is a good example of an endothermic reaction.
Question 10
Why is respiration considered an exothermic reaction?
Solution:
During respiration, glucose combines with oxygen in the cells of our body to form carbon dioxide and water along with the production of energy.
Glucose + oxygen → carbon dioxide + water + energy
It is an exothermic reaction, because energy is produced during this process.
Question 11
Why are decomposition reactions called the opposite of combination reactions? Write equations for decomposition reactions.
Solution:
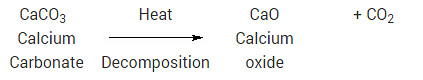
Essentially, decomposition reactions are the opposite of combination reactions. A compound decomposes (i.e., “splits-up”) into two or more compounds and/or elements. For example, mercury (II) oxide will, decompose into mercury metal and oxygen, upon heating:
2HgO → 2Hg + O2
Since heat had to be added to make this reaction occur, it is an endothermic reaction. Most decomposition reactions are endothermic. Another example of decomposition reaction is the heating of calcium carbonate (sea shells, chalk):
Question 12
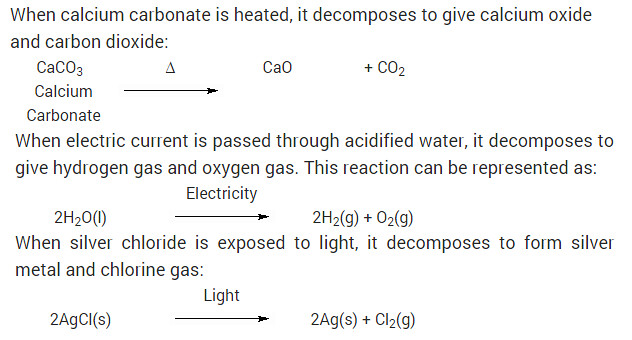
Write one equation each for decomposition reactions in which energy is supplied in the form of heat, light or electricity?
Solution:
When calcium carbonate is heated, it decomposes to give calcium oxide and carbon dioxide:
Question 13
What is the difference between displacement and double displacement reactions? Write relevant equations for the above?
Solution:
The difference between the displacement and double displacement reactions is that in a displacement reaction one element displaces another by virtue of it being more reactive whereas in a double displacement two anions and two cations switch places between two compounds respectively.
For example:
CuSO4 (aq) + Zn(s) → ZnSO4 + Cu(s)
Blue copper sulphate solution reacting with solid zinc will give rise to colourless zinc sulphate solution and solid copper. Thus Zn displaces Cu in the salt form. Zn is more reactive than Cu.
An example of double displacement reaction is the reaction between sodium carbonate and calcium chloride, both in aqueous
Solution:
Na2CO3 (aq) + CaCl2 (aq) → CaCO3 (ppt) + 2NaCl(aq)
Question 14
In the refining of silver, the recovery of silver from silver nitrate solution involves displacement by copper metal. Write down the reaction involved.
Solution:
![]()
Question 15
What do you mean by a precipitation reaction? Explain by giving examples:
Solution:
A precipitation reaction is a reaction in which soluble ions in separate solutions are mixed together to form an insoluble compound that settles out of solution as a solid. This insoluble compound is called a precipitate.
An example of a precipitation reaction
Aqueous silver nitrate (AgNO3), when added to a solution containing potassium chloride (KCl), precipitates a white solid, and silver chloride is observed.
AgNO3 (aq) + KCl(aq) → AgCl(s) + KNO3(aq)
The silver chloride (AgCl) has formed a solid, which is observed as a precipitate.
Question 16
A shiny brown colored element ‘X’ on heating in the air becomes black in color. Name the element ‘X’ and the black coloured compound formed.
Solution:
The shiny brown colored element X is copper metal (Cu). When copper metal is heated in air, it forms a black colored compound copper oxide. So, the black colored compound is the copper oxide or copper (II) oxide, CuO.
Question 17
Why do we apply paint on iron articles?
Solution:
Rust is a soft and porous substance, which gradually falls from the surface of an iron object, and then the iron below starts rusting. Thus, rusting of iron (or corrosion of iron) is a continuous process which, if not prevented in time, eats up the whole iron object. So, when we apply paint on iron articles it reduces the rusting of iron.
Question 18
Oil and fat containing food items are flushed with nitrogen. Why?
Solution:
Packaging fat and oil containing foods in nitrogen gas can prevent rancidity. When the fat and oil present in food materials gets oxidised (in air), their oxidation products have an unpleasant smell and taste. When it is surrounded by unreactive gas, nitrogen, there is no oxygen (of air) to cause its oxidation and make it rancid.
Question 19
Explain the following terms with one example each.
a. Corrosion
b. Rancidity
Solution:
a. Corrosion is the process in which metals are eaten up gradually by the action of air, moisture or a chemical on their surface. Corrosion is caused mainly by the oxidation of metals by the oxygen of air.
Example: Rusting of iron is the most common form of corrosion. When an iron object is left in damp air for a considerable period of time, it gets covered with a red-brown flaky substance called ‘rust’. This is called rusting of iron.
b. The condition produced by aerial oxidation of fat and oil in food which is marked by an unpleasant smell and taste is called rancidity.
Example: Rancidity can be retarded by keeping food in a refrigerator.
The refrigerator has a low temperature inside it. When the food is kept in a refrigerator, the oxidation of fat and oil in it is slowed down due to low temperature. So, the development of rancidity due to oxidation is retarded.
Question 20
Why should a magnesium ribbon be cleaned before burning in air?
Solution:
To remove the oxide layer and facilitates rapid burning.
Question 21
Write the balanced equation for the following chemical reactions.
i. Hydrogen + Chloride → Hydrogen chloride
ii. Barium chloride + Aluminium sulphate → Barium sulphate + Aluminium chloride
iii. Sodium + water → Sodium hydroxide + Hydrogen
Solution:
i. H2 + Cl2 → 2HCl
ii. 3BaCl2 + Al2(SO4)3 → 3BaSO4 + 2AlCl3
iii. 2Na + 2H2O → 2NaOH + H2
Question 22
Write a balanced chemical equation and state symbols for the following reactions.
a. Solutions of barium chloride and sodium sulphate in water react to give insoluble barium sulphate and the solution of sodium chloride
b. Sodium hydroxide solution (in water) reacts with hydrochloric acid solution (in water) to produce sodium chloride solution and water.
Solution:
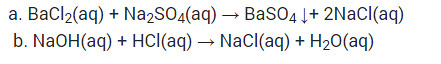
Question 23
A solution of a substance ‘X’ is used for white washing.
Name the substance ‘X’ and write its formula.
(i) Write the reaction of the substance ‘X; named in (ii) above with water
Solution:

Question 24
Why does the colour of copper sulphate solution change when an iron nail is dipped into it?
Solution:
In this reaction, iron displaces copper from copper sulphate solution. The deep blue colour of copper sulphate solution fades due to the formation of light green solution of iron sulphate. A red-brown coating of copper metal is formed on the surface of the iron metal. This displacement reaction occurs because iron is more reactive than copper.
Question 25
Identify the substances that are oxidised and the substances that are reduced in the following reactions.
i. 4Na(s) + O2 (g) → 2Na2O(s)
ii. CuO(s) + H2(g) → Cu(s) + H2O(l)
Solution:
i. 4Na(s) + O2 (g) → 2Na2O(s)
Here oxygen is added to sodium. The addition of oxygen is Called oxidation. So the substance that is oxidized is sodium Na.
ii. CuO(s) + H2(g) → Cu(s) + H2O(l)
In this reaction, copper oxide (CuO) gives the oxygen required for the oxidation of hydrogen; therefore, copper oxide is the oxidizing agent. Hydrogen is responsible for removing oxygen from copper oxide; therefore, hydrogen is the reducing agent here.
NCERT Solutions for Class 10 Science Chapter 1 (MCQs) [1 Mark each]
Question 1.
When crystals of lead nitrate are heated strongly in a dry test tube
(a) crystals immediately melt
(b) a brown residue is left
(c) white fumes appear in the test tube
(d) a yellow residue is left
Answer:
(b)Pungent smelling, brown fumes are evolved due to NO2 gas and brown coloured residue of lead oxide (PbO) is left.
![]()
Question 2.
A dilute ferrous sulphate solution was gradually added to the beaker containing acidified permanganate solution. The light purple colour of the solution fades and finally disappears. Which of the following is the correct explanation for the observation? [NCERT Exemplar]
(a) KMnO4 is an oxidising agent, it oxidises FeSO4
(b) FeSO4 acts as an oxidising agent and oxidises KMnO4
(c) The colour disappears due to dilution, no reaction is involved
(d) KMnO4 is an unstable compound and decomposes in the presence of FeSO4 to a colourless compound
Answer:
(a)Potassium permanganate (KMnO4) in the presence of dil. H2SO4, i.e. in acidic medium, acts as a strong oxidising agent. In acidic medium, KMnO4 oxidises ferrous sulphate to ferric sulphate.
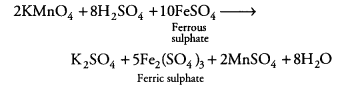
Question 3.
Dilute hydrochloric acid is added to granulated zinc taken in a test tube. The following observations are recorded. Point out the correct observation.
(a) The surface of metal becomes shining
(b) The reaction mixture turns milky
(c) Odour of a pungent smelling gas is recorded
(d) A colourless and odourless gas is evolved
Answer:
(d)Zinc metal reacts with dil. HCl to form zinc chloride and bubbles of colourless and odourless hydrogen gas is evolved.
![]()
Question 4.
When a magnesium ribbon is burnt in air, the ash formed is
(a) black
(b) white
(c) yellow
(d) pink
Answer:
(b)When a Mg ribbon is burnt in air, the ash formed is of magnesium oxide which is white in colour.
2Mg(r) + O2(g) → 2MgO(f)
Question 5.
Three beakers labelled as A, B and C each containing 25 mL of water were taken. A small amount of NaOH, anhyd. CuSO4 and NaCl were added to the beakers A, B and C, respectively. It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas, in case of beaker C, the temperature of the solution falls. Which one of the following statements is/are correct?
I. In beakers A and B, exothermic process has occurred.
II. In beakers A and B, endothermic process has occurred.
III. In beaker C, the exothermic process has occurred.
IV. In beaker C, endothermic process has occurred. [NCERT Exemplar]
(a) Only I
(b) Only II
(c) I and IV
(d) II and III
Answer:
(c)As in case of beakers A and B, heat is given out, so temperature became high, hence it is an exothermic reaction while in beaker C, heat is absorbed from water, so temperature falls, hence it is an endothermic process.
Question 6.
Which of the following will be required to identify the gas evolved when dilute hydrochloric acid reacts with zinc metal?
(a) Red litmus paper
(b) pH paper
(c) Lime water
(d) A burning splinter
Answer:
(d)On reacting with dil. HCl, zinc metal forms zinc chloride and hydrogen gas is evolved. Presence of hydrogen gas can be checked by a burning splinter because H4 gas burnt in a splinter with a pop sound.
![]()
Question 7.
On immersing an iron nail in CuSO4 solution for few minutes, you will observe that
(a) no reaction takes place
(b) the colour of solution fades away
(c) the surface of iron nails acquire a black coating
(d) the colour of solution changes to green
Answer:
(b)Fe(s) + CuSO4 (aq) → FeSO4 (aq) + Cu(s)
Fe is present above in the reactivity series of metals. Hence, Fe displaces Cu from CuSO4 solution. And the colour of the solution fades away. This is an example of displacement reaction.
Question 8.
What happens when ferrous sulphate crystals are heated? [CCE 2014]
(a) A gas having the smell of burning sulphur is evolved
(b) No gas is evolved
(c) Brown coloured gas is evolved
(d) Colourless and odourless gas is evolved
Answer:
(a)The green colour of ferrous sulphate crystals changes to brownish black ferric oxide and smell of burning sulphur is evolved due to SO2 and SO3.
![]()
Question 9.
The colour of the precipitate formed when barium chloride solution is mixed with sodium sulphate solution is [CCE 2014]
(a) blue
(b) black
(c) white
(d) green
Answer:
(c)This is an example of a double displacement reaction and a white precipitate of barium sulphate is formed.
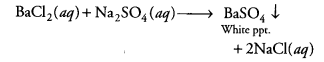
Question 10.
How the colour changes when the gases after thermal decomposition of ferrous sulphate come in contact with an acidified solution of potassium dichromate?
(a) Green to orange
(b) Red to colorless
(c) Orange to green
(d) Blue to green
Answer:
(c) The color changes from orange to green due to the formation of iron (III) sulphate
Chemical Reactions and Equations Chapter Wise Important Questions Class 10 Science
Short Answer Type Questions[l] [2 Marks] -Year 2015
1.“We need to balance a skeltal chemical equation.” Give reason to justify the statement.
Answer. Skeltal chemical equation are unbalanced. We need to balance chemical equation because of law of conservation of mass. It states that ‘matter can neither be created nor be destroyed’. Therefore chemical equation must be balanced in each and every chemical reaction.
2.Giving an example list two information which make a chemical equation more useful (informative).
Answer.
(i) Physical state of reactants must be mentioned, e.g.
2H2 (g) + O2 (g)——– > 2H20 (l)
(ii) Condition in which reaction takes place are written on the arrow head, e.g.
![]()
Consider the following chemical reaction
X + Barium chloride——–> Y + Sodium chloride
(White ppt)
(a) Identify ‘X’ and ‘Y’
(b) The type of reaction
(a) ‘X’ is Na2SO4 and Y is BaSO4.
(b) The type of reaction
Na2SO4 + BaCl2—–> BaSO4 + 2NaCl
(White ppt)
The reaction is precipitation reaction. It is also called double displacement reaction.
CBSE Class 10 Science – More Resources
5.(i) Write a balanced chemical equation for process of photosynthesis.4. Name the reducing agent in the following reaction:
3MnO2 + 4Al———— > 3Mn + 2Al2O3
State which is more reactive, Mn or A1 and why?
Answer. ‘Al’ is reducing agent.
‘AT is more reactive than Mn v ‘Al’ displaces Mn from its oxide.
(ii)When do desert plants take up carbon dioxide and perform photosynthesis?
Answer.
![]()
(ii) In desert plants the stomata are open at night. They take CO2 at night and is stored in the form of acid and is used during day time for photosynthesis.
Short Answer Type Questions[ll] [3 Marks] -Year 2015
6.A Name the type of chemical reaction represented by the following equation:

Answer.
(i) Combination reaction
(ii) Double displacement reaction (Precipitation reaction)
(iii) Decomposition reaction.
7. Write the chemical equation of the reaction in which the following changes have taken place with an example of each:
(i) Change in colour
(ii) Change in temperature
(iii) Formation of precipitate
Answer.
(i)Cu (s) + 2AgNO3 (aq)———–> Cu(NO3)2(aq) + 2Ag
The solution will become blue in colour and shiny silver metal will be deposited.
(ii) NaOH + HCl ———–> NaCl + H2O+ heat
The temperature will increase because heat will be evolved.
(iii) Pb(NO3)2 (aq) + 2KI (aq)———–> Pbl2 (s) + 2KNO3 (aq)
Yellow ppt
Yellow precipitate of Pbl2will be formed.
8.State the type of chemical reactions and chemical equations that take place in the following:
(i) Magnesium wire is burnt in air.
(ii) Electric current is passed through water.
(iii) Ammonia and hydrogen chloride gases’are mixed.
Answer.

9.(a) Write the essential condition for the following reaction to take place:
2AgBr—-> 2Ag + Br2
Write one application of this reaction.
(b) Complete the following chemical equation of a chemical reaction 2FeS04 —
![]()
(c) What happens when water is added to quick line. Write chemical equation.
Answer.
![]()
This reaction is used in photography.
![]()
(c) Slaked lime is formed with hissing sound and lot of heat is envolved.
10. 2g of ferrous sulphate crystals are heated in a dry boiling tube.
(i) List any two observations.
(ii) Name the type of chemical reaction taking place.
(iii) ‘Write the chemical equation for the reaction.
Answer.
(i) •Green colour of Fe SO4 disappears and reddish brown solid is formed.
• Smell of burning sulphur.
(ii) Decomposition reaction
![]()
Long Answer Type Questions [5 Marks] -Year 2015
11. (a) Define a balanced chemical equation. Why should an equation be balanced?
(b) Write the balanced chemical equation for the following reaction:
(i) Phosphorus burns in presence of chlorine to form phosphorus penta chloride.
(ii) Burning of natural gas.
(iii) The process of respiration.
Answer.
(a) Balanced chemical equation has an equal number of atoms of different elements in the reactants and products. According to law of conservation of mass, matter can neither be created nor be destroyed in a chemical reaction.
(b)(i) P4 (s) + 10Cl2 (g) ———> 4PCl5 (S)
(i)CH4 (g) + 2O2 (g) ———> CO2 (g) + 2H2O(l) + heat energy
(iii) C6H12O6 (s) + 6O2 (g) + 6H2O ———> 6CO2 (aq) + 12H2O (l) + energy
12.(a) Explain two ways by which food industries prevent rancidity.
(b) Discuss the importance of decomposition reaction in metal industry with three points.
Answer.
(a) (i) Rancidity can be prevented by adding antioxidants to food containing
fat and oil, e.g. butylated hydroxy anisole is added to butter as antioxidant.
(ii) It can be prevented by packaging fat and oil containing foods in nitrogen gas.
(b) (i) Molten NaCl is electrolytically decomposed to form sodium metal.
(ii) Aluminium metal is obtained by electric decomposition of bauxite ore mixed with cryolite.
(iii) Carbonate ores are thermally decomposed to give metal oxide which on reduction give metal.
Short Answer Type Question[I] [2 Marks] -Year 2014
13. What is observed when a solution of potassium iodide solution is added to a solution of lead nitrate? Name the type of reaction. Write a balanced chemical equation to represent the above chemical reaction.
Answer.Yellow precipitate of lead iodide is formed. It is precipitation reaction.
Pb( NO3)2 (aq) + 2KI (aq) —-> Pbl2 (s) + 2KNO3 (aq)
It is also called double displacement reaction.
short Answer Type Question[ll] [3 Marks] -Year 2014
14.Write chemical equation reactions taking place when carried out with the help of
(a) Iron reacts with steam
(b) Magnesium reacts with dil HCl
(c) Copper is heated in air.
Answer.

Long Answer Type Question [5 Marks] -Year 2014
15.(a) Write one example for each of decomposion reaction carried out with help of
(i) Electricity (ii) Heat (iii) Light
(b) Which of the following statements is correct and why copper can displace silver from silver nitrate and silver can displace copper from copper sulphate solution.
Answer.

Short Answer Type Questions[ll] [3 Marks] -Year 2013
16.Which products will be obtained when lead nitrate is heated simply. Write balanced chemical equation for the reaction? State the type of chemical reaction that occur in the change.
Answer. Lead monoxide, nitrogen dioxide and oxygen gas will be liberated.
![]()
It is thermal decomposition reaction.
17. What is meant by skeltal type chemical equation? What does it represent? Using the equation for electrolytic decomposition of water, differentiate between a skeltal chemical equation and a balanced chemical equation.
Answer. The equations in which gaseous are written in atomic form instead of molecular form and equation is not balanced, are called skeltal type equation. They represent gaseous elements formed in atomic state and equation is not balanced

Short Answer Type Questions[l] [2 Marks]-Year 2012
18.Write balanced chemical equations for the following reactions.
(i) Silver bromide on exposure to sunlight decomposes into silver and bromine,
(ii) Sodium metal reacts with water to form sodium hydroxide and hydrogen gas.
Answer.

19.Identify the type of reaction(s) in the following equations.
(i)CH4 + 2O2 CO2 + 2 H2O
(ii) Pb(NO3)2 + 2KI ——–>Pbl2 + 2KNOs
(iii) CaO + H2O ——–> Ca(OH)2
(iv) CuSO4 + Zn ——–> ZnSO4 + Cu
Answer.
(i) Combustion reaction and oxidation reaction.
(ii)Double displacement reaction and precipitation reaction.
(iii) Combination reaction.
(iv) Displacement reaction.
20.Write balanced equation for the reaction between magnesium and hydrochloric acid. Name the product obtained, identify the type of reaction.
Answer.
![]()
The product formed is magnesium chloride and hydrogen gas.
It is a displacement reaction.
21.Describe an activity to observe what happens when quick lime is added to water taken in a beaker. State two important observations and name the type of reaction taking place.
Answer.
Aim: To observe what happens when quicklime is added to water taken in a beaker.
Materials Required:- Quicklime (calcium oxide), water, beaker.

Procedure:
1. Take 5 g of calcium oxide in a beaker.
2. Add water to it slowly.
3. Touch the beaker.
4. Note down the observations.
Observation: Calcium oxide reacts with water
vigorously to form calcium hydroxide with the evolution of heat.
Chemical Reaction:

Conclusion: The reaction between CaO (Calcium oxide) and H2O is a combination reaction. It is an exothermic process because heat is evolved.
22.What is the colour of ferrous sulphate crystals? How does this colour change after heating?
Answer.The colour of ferrous sulphate is pale green. The colour changes to reddish brown on heating due to formation of iron (III) oxide.
Give an example each for thermal decomposition and photochemical decomposition reactions. Write relevant balanced chemical equations also.
Thermal decomposition reaction:

Photochemical decomposition reaction:
![]()
24. Why does the colour of copper sulphate solution change when an iron nail is dipped in it? Write two observations.
Answer. It is because displacement reaction takes place.
Iron displaces copper from copper sulphate solution and forms pale green
coloured solution of FeS04 and reddish brown copper metal gets deposited.
Fe(s) + CuS04(aq) ——–> FeS04(aq) + Cu(s)
25. Translate the following statement into chemical equation and then balance it Barium chloride reacts with aluminium sulphate to give aluminium chloride and a precipitate of barium sulphate. State the two types in which this reaction can be classified.
Answer. 3BaCl2(aq) + A12(S04)3(aq) ——–> 3BaS04(s) + 2AlCl3(aq)
It can be classified as double displacement as well as precipitation reaction.
26. Why are decomposition reactions called the opposite of combination reactions? Write equations for these reactions.
Answer. In decomposition reaction, a compound is broken down into simpler compounds or elements, e.g.
![]()
Combination reaction is a reaction in which two or more elements or compounds combine to form a new compound, e.g.
![]()
Thus, decomposition and combination reactions are opposite to each other.
Short Answer Type Questions[ll] [3 Marks] -Year 2012
27. The following diagram displays a chemical reaction. Observe carefully and answer the following questions

(a) Identify the type of chemical reaction that will take place and define it. How will the colour of the salt change?
Write the chemical equation of the reaction that takes place.
(c) Mention one commercial use of this salt.
Answer. (a) Photochemical decomposition reaction: Those reactions in which a compound breaks down into simple substances in presence of light are called photochemical decomposition reaction. The colour of salt will change from white to grey.
![]()
(c) Silver chloride is used in photography.
28. What is rancidity? Mention any two ways by which rancidity can be prevented.
Answer. The process in which taste and smell of food gets spoiled is called rancidity. It happens due to oxidation.
Prevention from rancidity:
(i) Antioxidants are added to fatty acids to prevent oxidation, e.g. chips are packed in presence of nitrogen gas which prevents spoilage by oxidation.
(ii)Food should be kept in airtight container in refrigerator.
29.Write balanced chemical equation for the reactions that take place during respiration. Identify the type of combination reaction that takes place during this process and justify the name. Give one more example of this type of reaction.
Answer. CgH1206 + 6O2 —————> 6CO2 + 6H20 + heat
It is an exothermic combination reaction because heat is evolved.
CH4(g) + 2O2(g) ————–>CO2 (g) + 2H20
Combustion of methane is another example of exothermic combination reaction.
30. What is redox reaction? Identify the substance oxidised and the substance reduced in the following reactions.
(i)2PbO + C —–> 2Pb + CO2
(ii)MnO2 + 4HCl —–> MnCl2 + 2H20 + Cl2
Answer. Those reactions in which oxidation and reduction takes place simultaneously are called redox reactions.
(i) PbO is getting reduced and C is getting oxidised.
(ii) MnOs is getting reduced and HCl is getting oxidised.
31. Write the balanced chemical equations for the following reactions and identify the type of reaction in each case.
Thermite reaction, iron (III) oxide reacts with aluminium and gives molten iron and aluminium oxide.
Answer.

It is a displacement reaction because A1 is displacing Fe from Fe2O3.
Molten iron is used’for repairing broken railway tracks.
32. A solution of potassium chloride when mixed with silver nitrate solution, an insoluble white substance is formed. Write the chemical reaction involved and also mention the type of the chemical reaction?
Answer.
 .
.
It is a double displacement reaction. It is also a precipitation reaction as AgCl is a white precipitate.
Very Short Answer Type Questions [1 Mark] -Year 2011
33.State one basic difference between a physical change and a chemical change.
Answer. In physical change, no new substance is formed, whereas in a chemical change, new substance(s) is/are formed.
34 What is meant by a chemical reaction?
Answer. The reaction representing a chemical change is called a chemical reaction.
35.AgN03(aq) + NaCl(aq)——————– > AgCl(s)4↓ + NaN03(aq)
FeS + H2S04————- > FeS04 + H2S↑
Consider the above mentioned two chemical equations with two different kinds of arrows (↑and ↓) along with product. What do these two different arrows indicate?
Ans,↑shows the gas is evolved whereas ↓shows insoluble substance (precipitate) is formed.
36. Hydrogen being a highly inflammable gas and oxygen being a supporter of combustion, yet water which is a compound made up of hydrogen and oxygen is used to extinguish fire. Why?
Answer. It is because properties of compound (H2O) are different from properties of its constituting elements, i.e. H2and O2.
Short Answer Type Questions[l] [2 Marks] -Year 2011
37.Using a suitable chemical equation, justify that some chemical reactions are determined by:
(i) change in colour, (ii) change in temperature.
Answer.

38.(a) A solution of substance ‘X’ is used for white washing. What is the substance ‘X’? State the chemical reaction of ‘X’ with water.
(b) Why does the colour of copper sulphate solution change when an iron nail is dipped in it?
Answer.
(a) ‘X’ is calcium oxide (CaO).
CaO(s) + H2O(l) —–> Ca(OH)2(aq) + heat
(a) It is because iron displaces copper from CuS04 to form FeS04 which is pale green.
Fe(s) + CUS04 (aq)—–> FeS04(aq) + Cu(s)
Blue Pale green
39.Balance the following chemical equations.

Answer.

40.Write the balanced equation for the. following reaction and identify the type of reaction in each case.
(i) Potassium bromide + Barium iodide—-> Potassium iodide + Barium bromide.
(ii) Hydrogen(g) + Chlorine(g)—-> Hydrogen chloride(g)
Answer.

41. A zinc plate was put into a solution of copper sulphate kept in a glass container. It was found that blue colour of the solution gets fader and fader with the passage of time. After few days, when zinc plate was taken out of the solution, a number of holes were observed on it.
(i) State the reason for changes observed on the zinc plate.
(ii) Write the chemical equation for the reaction involved.
Answer.
(i) It is because zinc has displaced copper from CuS04. Zinc metal has been used to form zinc sulphate, therefore, number of holes were observed.
![]()
42. A white salt on heating decomposes to give brown fumes and a residue is left behind.
(i) Name the salt.
(ii) Write the equation for the decom-position reaction.
Answer.
(i) Lead nitrate is white salt.
![]()
43. When a solution of potassium iodide is added to a solution of lead nitrate in a test tube, a reaction takes place.
(a) What type of reaction is this?
(b) Write a balanced chemical equation to represent the above reaction.
Answer.
(a) Double displacement as well as precipitation reaction.

44. Define combination reaction. Give one example of a combination reaction which is also exothermic.
Answer. A reaction in which two elements or compounds combine to form a single compound is called combination reaction.
![]()
It is also an exothermic reaction along with a combination reaction because heat is evolved.
Short Answer Type Questions[ll] [3 Marks]
45.(a) Classify the following reactions into different types.

(b) Which of the above reaction(s) is/are precipitation reaction(s)? Why is a reaction called precipitation reaction?
Answer.
(a) (i) Precipitation reaction (Double displacement reaction)
(ii) Combination reaction (in) Decomposition reaction
(b) Reaction (i) is a precipitation reaction because one of the product formed is insoluble in water.
46. Write balanced equations for the following mentioning the type of reaction involved.
(i) Aluminium + Bromine —–> Aluminium bromide
(ii) Calcium carbonate—–> Calcium oxide + Carbon dioxide
(iii) Silver chloride—–>Silver + Chlorine
Answer.

47.(a) Why is respiration considered as an exothermic reaction?
(b) Define the terms oxidation and reduction.
(c) Identify the substance that is oxidised and reduced in the following reaction.
![]()
Answer. (a) It is because heat is evolved during respiration.
(b) Oxidation is a process in which O2 is added or H2 is removed or loss of electrons take place. Reduction is a process in which H2 is added or O2. is removed or gain of electrons take place.
(c) Zn is getting oxidised, CuO is getting reduced.
48. What is meant by
(i) precipitation reaction,
(ii) exothermic reaction,
(iii) oxidation reaction?
Write balanced chemical equations for an example of each.
Answer.(i) Precipitation reaction: The reaction in which two compounds exchange their
ions and the product formed is insoluble in water is called precipitation reaction.
![]()
(ii) Exothermic reaction: The reaction in Which heat is evolved is known as exothermic reaction.
![]()
(iii) Oxidation reaction: The reaction in which Og is added or H2 is removed or loss of electrons takes place is called oxidation reaction.
![]()
49.You might have noted that when copper powder is heated in a china dish, the surface of copper powder becomes coated with a black colour substance.
(i) How has this black coloured substance formed?
(ii) What is that black substance?
(iii) Write the chemical equation of the reaction that takes place.
Answer.
(i) Copper reacts with oxygen to form copper oxide which is black, i.e. oxidation of copper takes place.
(ii)Copper oxide
![]()
Very Short Answer Type Questions [1 Mark] -Year 2010
50. What happens chemically when quicklime is added to water filled in a bucket?
Answer. Quicklime reacts with water to form slaked lime and produces lot of heat and hissing sound.

51. On what basis is a chemical equation balanced?
Answer. A chemical reaction is balanced on the basis of law of conservation of mass.
52. What change in colour is observed when white silver chloride is left exposed to sunlight? State the type of chemical reaction in this change.
Answer. Silver chloride becomes grey. It is a photochemical decomposition reaction.
53. Write a balanced chemical equation for the reaction between sodium chloride
and silver nitrate indicating the physical state of the reactants and the products.
Answer.
![]()
Short Answer Type Questions[l] [2 Marks]
54. What happens when an aqueous solution of sodium sulphate reacts with an aqueous solution of barium chloride? State the physical conditions of reactants in which the reaction between them will not take place. Write the balanced chemical equation for the reaction and name the type of reaction.
Answer. White precipitate of barium sulphate is formed.
If both reactants are in solid state, then the reaction will not take place between them.
![]()
It is a double displacement as well as a precipitation reaction.
55. What is a redox reaction? When a magnesium ribbon burns in air with a dazzling flame and forms a white ash, is magnesium oxidised or reduced? Why?
Answer. The reactions in which oxidation (loss of electrons) and reduction (gain of electrons) take place simultaneously are called redox reactions.

Magnesium is getting oxidised because it is losing electrons to form Mg2+ and oxygen is gaining electrons to form O2-, therefore it is getting reduced.
56. Write any two observations in an activity which may suggest that a chemical reaction has taken place. Give an example in support of your answer.
Answer. Any two of these observations will suggest chemical reaction has taken place.
(i) Change in state.
(ii)Change in colour.
(iii) Evolution of gas.
(iv)Change in temperature.
For example, lead nitrate is white crystalline solid which on heating gives yellowish brown solid (lead monoxide). A brown gas and a colourless gas is also evolved. It shows chemical reaction has taken place.

57.When the powder of a common metal is heated in an open china dish, its colour turns black. However, when hydrogen is passed over the hot black substance so formed, it regains its original colour. Based on the above information, answer the following questions.
(i) What type of chemical reaction takes place in each of the two given steps?
(ii) Name the metal initially taken in the powder form. Write balanced chemical equations for both reactions.
Answer.
(i) In first step, oxidation takes place.In second step, redox reaction takes place.
(ii)Metal in the powder form is copper.

Very Short Answer Type Questions [1 Mark] -Year 2009
58.In electrolysis of water, why is the volume of gas collected over one electrode double that of gas collected over the other electrode?
Answer.It is because water contains hydrogen and oxygen in the ratio of 2 : 1.
59.Balance the following chemical equations.
Answer.

Short Answer Type Questions[l] [2 Marks] -Year 2009
60. Name the products formed on strongly heating ferrous sulphate crystals. What type of chemical reaction occurs in this change?
Answer.

It is decomposition reaction.
61. What is an oxidation reaction? Give an example of oxidation reaction. Is oxidation an exothermic or an endothermic reaction?
Answer. The reaction in which oxygen or electronegative element is added or hydrogen or electropositive element is removed or loss of electrons takes place, is called an oxidation reaction, e.g. ,

Oxidation reactions are mostly exothermic in nature because heat is evolved in this process.
62. Describe an activity to demonstrate the change that takes place when white silver chloride is kept in sunlight. State the type of chemical reaction which takes place.
Answer.
Aim: To demonstrate the change that takes place when white silver chloride is kept in sunlight.
Materials Required: AgNO3(aq), NaCl(aq), test tubes.

Procedure:
1. Take 5 ml of silver nitrate solution in a test tube.
2. Prepare sodium chloride solution in another test tube.
3. Add sodium chloride solution into test tube containing silver nitrate solution.
4. Observe the colour of silver chloride formed chloride to grey silver metal Dry it with the help of filter papers and place it on the watch glass.
5. Place the watch glass under sunlight for sometime.
6. Observe the colour of the silver chloride after sometime. Observation: White silver chloride turns grey in sunlight because silver metal is formed.

Explanation: Silver chloride is photosensitive. It decomposes in presence of sunlight to form silver metal and chlorine gas.
Conclusion: Decomposition of silver chloride in presence of sunlight is photochemical decomposition reaction.
63.When magnesium ribbon burns in air or oxygen, a product is formed. State the type of chemical reaction and name the product formed in the reaction. Write balanced chemical equation of this reaction.
Answer.
![]()
The type of reaction is combination reaction and the product formed is magnesium oxide.
64.Distinguish between a displacement reaction and a double displacement reaction. Identify the displacement and the double displacement reaction from the following reactions.
Answer.

Displacement reaction is a reaction in which more reactive metal can displace less reactive metal from its salt solution.
Double displacement reaction are those reactions in which compounds exchange their ions to form two new compounds (?) Double displacement reaction (ii) Displacement reaction
65.When you have mixed the solutions of lead(II) nitrate and potassium iodide,
(i) what was the colour of the precipitate formed and can you name the precipitate?
(ii) write the balanced chemical equation for this reaction.
(iii) is this also a double displacement reaction?
Answer.
(i) The colour of the precipitate is yellow. The name of compound formed as a precipitate is Pbl2 (lead iodide).
![]()
(iii) Yes, it is also a double displacement reaction.
66.What do you mean by exothermic and endothermic reactions? Give examples.
Answer.Exothermic reactions are those in which heat is evolved, e.g.

Endothermic reactions are those reactions in which heat is absorbed, e.g.

CBSE Class 10 Science Notes Chapter 1 Chemical Reactions and Equations
Chemical Reactions and Equations: Balanced and unbalanced chemical equations and balancing of chemical equations.
What is a chemical reaction Class 10?
Chemical Reaction: The transformation of chemical substance into another chemical substance is known as Chemical Reaction. For example: Rusting of iron, the setting of milk into curd, digestion of food, respiration, etc.
In a chemical reaction, a new substance is formed which is completely different in properties from the original substance, so in a chemical reaction, a chemical change takes place.
Only a rearrangement of atoms takes place in a chemical reaction.
- The substances which take part in a chemical reaction are called reactants.
- The new substances produced as a result of a chemical reaction are called products.
Example: The burning of magnesium in the air to form magnesium oxide is an example of a chemical reaction.
2Mg(s) + O2(g) △→ 2MgO(s)
Before burning in air, the magnesium ribbon is cleaned by rubbing with sandpaper.
This is done to remove the protective layer of basic magnesium carbonate from the surface of the magnesium ribbon.
Reactant: Substances which take part in a chemical reaction are called reactants.
Example: Mg and O2.
Product: New substance formed after a chemical reaction is called a product.
Example: MgO.
Characteristics of Chemical Reactions :
(i) Evolution of gas: The chemical reaction between zinc and dilute sulphuric acid is characterised by the evolution of hydrogen gas.
Zn(s) + H2SO4(aq) → ZnSO4(aq) + H2(g) ↑
(ii) Change in Colour: The chemical reaction between citric acid and purple coloured potassium permanganate solution is characterised by a change in colour from purple to colourless.
The chemical reaction between sulphur dioxide gas and acidified potassium dichromate solution is characterized by a change in colour from orange to green.
(iii) Change in state of substance: The combustion reaction of candle wax is characterised by a change in state from solid to liquid and gas (because the wax is a solid, water formed by the combustion of wax is a liquid at room temperature whereas, carbon dioxide produced by the combustion of wax is a gas). There are some chemical reactions which can show more than one characteristics.
(iv) Change in temperature: The chemical reaction between quick lime water to form slaked lime is characterized by a change in temperature (which is a rise in temperature).
The chemical reaction between zinc granules and dilute sulphuric acid is also characterised by a change in temperature (which is a rise in temperature).
(v) Formation of precipitate: The chemical reaction between sulphuric acid and barium chloride solution is characterised by the formation of a white precipitate of barium sulphate.
BaCl2(aq) + H2SO4(aq) → BaSO4(s) (ppt) + 2HCl(aq)
What is a chemical Equation Class 10?
Chemical Equation: Representation of chemical reaction using symbols and formulae of the substances is called Chemical Equation.
Example: A + B → C + D
In this equation, A and B are called reactants and C and D are called the products. The arrow shows the direction of the chemical reaction. Condition, if any, is written generally above the arrow.
When hydrogen reacts with oxygen, it gives water. This reaction can be represented by the following chemical equation:
Hydrogen + Oxygen → Water
H2 + O2 → H2O
In the first equation, words are used and in second, symbols of substances are used to write the chemical equation. For convenience, the symbol of substance is used to represent chemical equations.
A chemical equation is a way to represent the chemical reaction in a concise and informative way.
A chemical equation can be divided into two types: Balanced Chemical Equation and Unbalanced Chemical Equation.
(a) Balanced Chemical Equation: A balanced chemical equation has the number of atoms of each element equal on both sides.
Example: Zn + H2SO4 → ZnSO4 + H2
In this equation, numbers of zinc, hydrogen and sulphate are equal on both sides, so it is a Balanced Chemical Equation.
According to the Law of Conservation of Mass, mass can neither be created nor destroyed in a chemical reaction. To obey this law, the total mass of elements present in reactants must be equal to the total mass of elements present in products.
(b) Unbalanced Chemical Equation: If the number of atoms of each element in reactants is not equal to the number of atoms of each element present in the product, then the chemical equation is called Unbalanced Chemical Equation.
Example: Fe + H2O → Fe3O4 + H2
In this example, a number of atoms of elements are not equal on two sides of the reaction. For example; on the left-hand side only one iron atom is present, while three iron atoms are present on the right-hand side. Therefore, it is an unbalanced chemical equation.
Balancing a Chemical Equation: To balance the given or any chemical equation, follow these steps:
Fe + H2O → Fe3O4 + H2
Write the number of atoms of elements present in reactants and in products in a table as shown here.
| Name of atom | No. of atoms in the reactant | No. of atoms in the product |
| Iron | 1 | 3 |
| Hydrogen | 2 | 2 |
| Oxygen | 1 | 4 |
Balance the atom which is maximum in number on either side of a chemical equation.
In this equation, the number of oxygen atom is the maximum on the RHS.
To balance the oxygen, one needs to multiply the oxygen on the LHS by 4, so that, the number of oxygen atoms becomes equal on both sides.
Fe + 4 × H2O → Fe3O4 + H2
Now, the number of hydrogen atoms becomes 8 on the LHS, which is more than that on the RHS. To balance it, one needs to multiply the hydrogen on the RHS by 4.
Fe + 4 × H2O → Fe3O4 + 4 × H2
After that, the number of oxygen and hydrogen atoms becomes equal on both sides. The number of iron is one on the LHS, while it is three on the RHS. To balance it, multiply the iron on the LHS by 3.
3 × Fe + 4 × H2O → Fe3O4 + 4 × H2
Now the number of atoms of each element becomes equal on both sides. Thus, this equation becomes a balanced equation.
| Name of atom | No. of atoms in the reactant | No. of atoms in the product |
| Iron | 3 | 3 |
| Hydrogen | 8 | 8 |
| Oxygen | 4 | 4 |
After balancing, the above equation can be written as follows:
3Fe + 4H2O → Fe3O4 + 4H2.
To Make Equations More Informative:
Writing the symbols of physical states of substances in a chemical equation:
By writing the physical states of substances, a chemical equation becomes more informative.
- Gaseous state is represented by symbol (g).
- Liquid state is represented by symbol (l).
- Solid state is written by symbol (s).
- Aqueous solution is written by symbol (aq).
- Writing the condition in which reaction takes place: The condition is generally written above and/or below the arrow of a chemical equation.
Thus, by writing the symbols of the physical state of substances and condition under which reaction takes place, a chemical equation can be made more informative.
What are the types of a chemical reaction Class 10?
Types of Chemical Reactions: Combination Reaction, Decomposition Reaction, Displacement Reaction, Double Displacement Reaction, Neutralization Reactions, Exothermic – Endothermic Reactions and Oxidation-Reduction Reactions.
Types of Chemical Reactions:
Chemical reactions can be classified in following types:
(i) Combination Reaction: Reactions in which two or more reactants combine to form one product are called Combination Reactions.
A general combination reaction can be represented by the chemical equation given here:
A + B → AB
Examples:
When magnesium is burnt in the air (oxygen), magnesium oxide is formed. In this reaction, magnesium is combined with oxygen.
Mg(s) + O2(g) → 2MgO(s)
Magnesium + Oxygen → Magnesium Oxide
When carbon is burnt in oxygen (air), carbon dioxide is formed. In this reaction, carbon is combined with oxygen.
C (s) + O2(g) → CO2(g)
Carbon + Oxygen → Carbon dioxide
(ii) Decomposition Reaction: Reactions in which one compound decomposes in two or more compounds or elements are known as Decomposition Reaction. A decomposition reaction is just the opposite of combination reaction.
A general decomposition reaction can be represented as follows :
AB → A + B
Examples:
When calcium carbonate is heated, it decomposes into calcium oxide and carbon dioxide.
CaCO3(s) heat−→− CaO(s) + CO2(g)
Calcium carbonate → Calcium oxide + Carbon dioxide
When ferric hydroxide is heated, it decomposes into ferric oxide and water
2Fe(OH)3(s) △→ Fe2O3(s) + 3H2O(l)
Thermal Decomposition: The decomposition of a substance on heating is known as Thermal Decomposition.
Example: 2Pb(NO3)2(s) heat−→− 2PbO(s) + 4NO2(g) + O2(g)
Electrolytic Decomposition: Reactions in which compounds decompose into simpler compounds because of passing of electricity, are known as Electrolytic Decomposition. This is also known as Electrolysis.
Example: When electricity is passed in water, it decomposes into hydrogen and oxygen.
Photolysis or Photo Decomposition Reaction: Reactions in which a compound decomposes because of sunlight are known as Photolysis or Photo Decomposition Reaction.
Example: When silver chloride is put in sunlight, it decomposes into silver metal and chlorine gas.
2AgCl(s) (white) Sunlight−→−−−−− 2Ag(s) (grey) + Cl2(g)
Photographic paper has a coat of silver chloride, which turns into grey when exposed to sunlight. It happens because silver chloride is colourless while silver is a grey metal.
(iii) Displacement Reaction: The chemical reactions in which a more reactive element displaces a less reactive element from a compound is known as Displacement Reactions. Displacement reactions are also known as Substitution Reaction or Single Displacement/ replacement reactions.
A general displacement reaction can be represented by using a chemical equation as follows :
A + BC → AC + B
Displacement reaction takes place only when ‘A’ is more reactive than B. If ‘B’ is more reactive than ‘A’, then ‘A’ will not displace ‘C’ from ‘BC’ and reaction will not be taking place.
Examples:
When zinc reacts with hydrochloric acid, it gives hydrogen gas and zinc chloride.
Zn(s) + 2HCl(aq) → ZnCl2(aq) + H2(g)
When zinc reacts with copper sulphate, it forms zinc sulphate and copper metal.
Zn(s) + CuSO4(aq) → ZnSO4(aq) + Cu(s)
(iv) Double Displacement Reaction: Reactions in which ions are exchanged between two reactants forming new compounds are called Double Displacement Reactions.
AB + CD → AC + BD
Examples:
When the solution of barium chloride reacts with the solution of sodium sulphate, white precipitate of barium sulphate is formed along with sodium chloride.
BaCl2(aq) + Na2SO4(aq) → BaSO4(s) (Precipitate) + 2NaCl(aq)
When sodium hydroxide (a base) reacts with hydrochloric acid, sodium chloride and water are formed.
NaOH(aq) + HCl(aq) → NaCl(aq) + H2O(l)
Note: Double Displacement Reaction, in which precipitate is formed, is also known as precipitation reaction. Neutralisation reactions are also examples of double displacement reaction.
Precipitation Reaction: The reaction in which precipitate is formed by the mixing of the aqueous solution of two salts is called Precipitation Reaction.
Example:

Neutralization Reaction: The reaction in which an acid reacts with a base to form salt and water by an exchange of ions is called Neutralization Reaction.
Example:

(v) Oxidation and Reduction Reactions:
Oxidation: Addition of oxygen or non-metallic element or removal of hydrogen or metallic element from a compound is known as Oxidation.
Elements or compounds in which oxygen or non-metallic element is added or hydrogen or metallic element is removed are called to be Oxidized.
Reduction: Addition of hydrogen or metallic element or removal of oxygen or non-metallic element from a compound is called Reduction.
The compound or element which goes under reduction in called to be Reduced.
Oxidation and Reduction take place together.
Oxidizing agent:
- The substance which gives oxygen for oxidation is called an Oxidizing agent.
- The substance which removes hydrogen is also called an Oxidizing agent.
Reducing agent:
- The substance which gives hydrogen for reduction is called a Reducing agent.
- The substance which removes oxygen is also called a Reducing agent.
The reaction in which oxidation and reduction both take place simultaneously is called Redox reaction.
When copper oxide is heated with hydrogen, then copper metal and hydrogen are formed.
CuO + H2 → Cu + H2O
(i) In this reaction, CuO is changing into Cu. Oxygen is being removed from copper oxide. Removal of oxygen from a substance is called Reduction, so copper oxide is being reduced to copper.
(ii) In this reaction, H2 is changing to H2O. Oxygen is being added to hydrogen. Addition of oxygen to a substance is called Oxidation, so hydrogen is being oxidised to water.
- The substance which gets oxidised is the reducing agent.
- The substance which gets reduced is the oxidizing agent.
(vi) Exothermic and Endothermic Reactions:
Exothermic Reaction: Reaction which produces energy is called Exothermic Reaction. Most of the decomposition reactions are exothermic.
Example:
Respiration is a decomposition reaction in which energy is released.

When quick lime (CaO) is added to water, it releases energy.

Endothermic Reaction: A chemical reaction in which heat energy is absorbed is called Endothermic Reaction.
Example: Decomposition of calcium carbonate.

Effects of Oxidation Reactions in Everyday life: Corrosion and Rancidity.
Corrosion: The process of slow conversion of metals into their undesirable compounds due to their reaction with oxygen, water, acids, gases etc. present in the atmosphere is called Corrosion.
Example: Rusting of iron.
Rusting: Iron when reacts with oxygen and moisture forms red substance which is called Rust.

The rusting of iron is a redox reaction.
Corrosion (rusting) weakens the iron and steel objects and structures such as railings, car bodies, bridges and ships etc. and cuts short their life.
Methods to Prevent Rusting
- By painting.
- By greasing and oiling.
- By galvanisation.
Corrosion of Copper: Copper objects lose their lustre and shine after some time because the surface of these objects acquires a green coating of basic copper carbonate, CuCO3.Cu(OH)2 when exposed to air.

Corrosion of Silver Metal: The surface of silver metal gets tarnished (becomes dull) on exposure to air, due to the formation of a coating of black silver sulphide(Ag2S) on its surface by the action of H2S gas present in the air.

Rancidity: The taste and odour of food materials containing fat and oil changes when they are left exposed to air for a long time. This is called Rancidity. It is caused due to the oxidation of fat and oil present in food materials.
Methods to prevent rancidity:
- By adding anti-oxidant.
- Vacuum packing.
- Replacing air by nitrogen.
- Refrigeration of foodstuff.
1. Chemical Reaction: During chemical reactions, the chemical composition of substances changes or new substances are formed.
2. Chemical Equation: Chemical reactions can be written in chemical equation form which should always be balanced.
3. Types of Chemical Reactions:
Combination reaction: A single product is formed from two or more reactants.
2Mg + O2 → 2MgO
Decomposition reaction: A single reactant breaks down to yield two or more products.
- Thermal decomposition: 2Pb(NO2)2 → 2PbO + 4NO2 + O2
- Electrolysis: 2H20 → 2H2 + O2
- Photochemical reaction: 2AgBr → 2Ag + Br2
Displacement reaction: One element is displaced by another element.
Zn + CuSO4 → ZnSO4 + Cu
Double displacement reaction: Exchange of ions between reactants.
AgNO3 + NaCl → AgCl + NaNO3
Redox reaction: Both oxidation and reduction take place simultaneously.
CuO + H2 → Cu + H2O
Exothermic reaction: A chemical reaction in which heat energy is evolved.
C + O2 → CO2 (g) + heat
Endothermic reaction: A chemical reaction in which heat energy is absorbed.
ZnCO3 + Heat → ZnO + CO2
Redox reaction: Chemical reaction in which both oxidation and reduction take place simultaneously.
4. Oxidation: Reaction that involves the gain of oxygen or loss of hydrogen.
5. Reduction: Reaction that shows the loss of oxygen or gain of hydrogen.
ZnO + C → Zn + CO
ZnO is reduced to Zn—reduction. C is oxidized to CO—Oxidation.
6. Effects of Oxidation Reactions in Our Daily Life:
- Corrosion: It is an undesirable change that occurs in metals when they are attacked by moisture, air, acids and bases.
Example, Corrosion (rusting) of Iron: Fe2O3. nH2O (Hydrated iron oxide) - Rancidity: Undesirable change that takes place in oil containing food items due to the oxidation of fatty acids.
Preventive methods of rancidity: Adding antioxidants to the food materials, storing food in the airtight container, flushing out air with nitrogen gas and refrigeration.
Chemical Reactions and Equation – CBSE Class 10 Science – Extra Questions
Question-1
What is the method of balancing chemical equation?
Solution:
Hit and trial method is used for balancing simple chemical equations. In this method, coefficients before the symbols/formulae of the reactants and products are adjusted in such a way that the total number of atoms of each element on both the sides becomes equal.
Question-2
Why is it necessary to balance a chemical equation?
Solution:
We know that, according to the law of conservation of mass, the total mass of all the atoms present in the reactants should be equal to that of all the atoms present in the products. This law will hold good only when the number of atoms of all types of elements on both sides is equal. Thus balancing of chemical equation is necessary so that the law of conservation of mass may be obeyed.
Question-3
In the equations given below, state giving reasons, whether substances have been oxidised or reduced.
(i) PbO + CO –> Pb + CO2
(ii) H2S + Cl2–>2HCl + S.
Solution:
(i) Carbon monoxide is oxidised as it gains oxygen.
(ii) Chlorine is reduced as it gains hydrogen.
Question-4
A sample of water weed was placed in water and exposed to sunlight. Bubbles of a gas are seen on the surface of the leaves.
(i) Name the gas evolved.
(ii) Name the process taking place.
(iii) Write a balanced equation of reaction taking place.
Solution:
(i) Oxygen
(ii) Photosynthesis
(iii) 6CO2 + 6H2O —> C6H12O6 + 6O2.
Question-5
Give balanced equations, wherever possible, or where this is not possible, explain the following by means of examples:
i) A reaction which gives out heat.
ii) A reaction which takes place with the help of sunlight.
iii) A reaction which is brought about by electric current.
iv) A reversible reactions.
(v) A reaction with a solid and gas which produces heat.
Solution:

Question-6
Balance the following chemical equations:

Question-7
Write the uses of decomposition reactions.
Solution:
The decomposition reactions carried out by electricity are used to extract several metals from their naturally occurring compounds like chlorides and oxides.
When passing electricity decomposes the fused metal chloride or metal oxide, then metal is produced at the cathode.
Question-8
What are the characteristics of chemical reactions?
Solution:
1. Evolution of a gas
2. Formation of a precipitate
3. Change in colour
4. Change in temperature
5. Change in state.
Question-9
Can rancidity retarded by storing foods away from light?
Solution:
In the absence of light, the oxidation of fats and oils present in food is slowed down and hence the development of rancidity is retarded.
Question-10
What are the different ways can make more informative about the chemical equation?
Solution:
1. By indicating the “physical states” of the reactants and products.
2. By indicating the “heat changes” taking place in the reaction.
3. By indicating the “conditions” under which the reaction takes place
